The Dream of a Diabetes Cure Gets Real
Imagine if people with type 1 diabetes could skip the daily insulin shots and finger pricks forever. That's not science fiction anymore—it's getting closer, thanks to some clever Swedish scientists who figured out how to grow functional insulin cells in a petri dish.
Type 1 diabetes happens when your immune system basically turns traitor and destroys the cells in your pancreas that make insulin. Without insulin, your body can't tell your cells to absorb glucose from your blood, which gets messy pretty fast. For decades, scientists thought, "Hey, why don't we just grow new insulin cells and pop them back in?" Sounds simple, right? It's not.
Why Growing Insulin Cells Is Harder Than It Sounds
Here's the thing: making functional insulin cells from stem cells sounds straightforward on paper, but it's been weirdly inconsistent in practice. Previous attempts would produce a jumble of different cell types—some useful, some not so much. The cells that did become insulin producers often weren't mature enough to do their job properly. It's like trying to train a student to be a surgeon, but half the students decided to become accountants instead, and the ones who stayed didn't know what they were doing yet.
The team at Karolinska Institutet decided to completely rethink the recipe.
A Better Way to Cook Up Insulin Cells
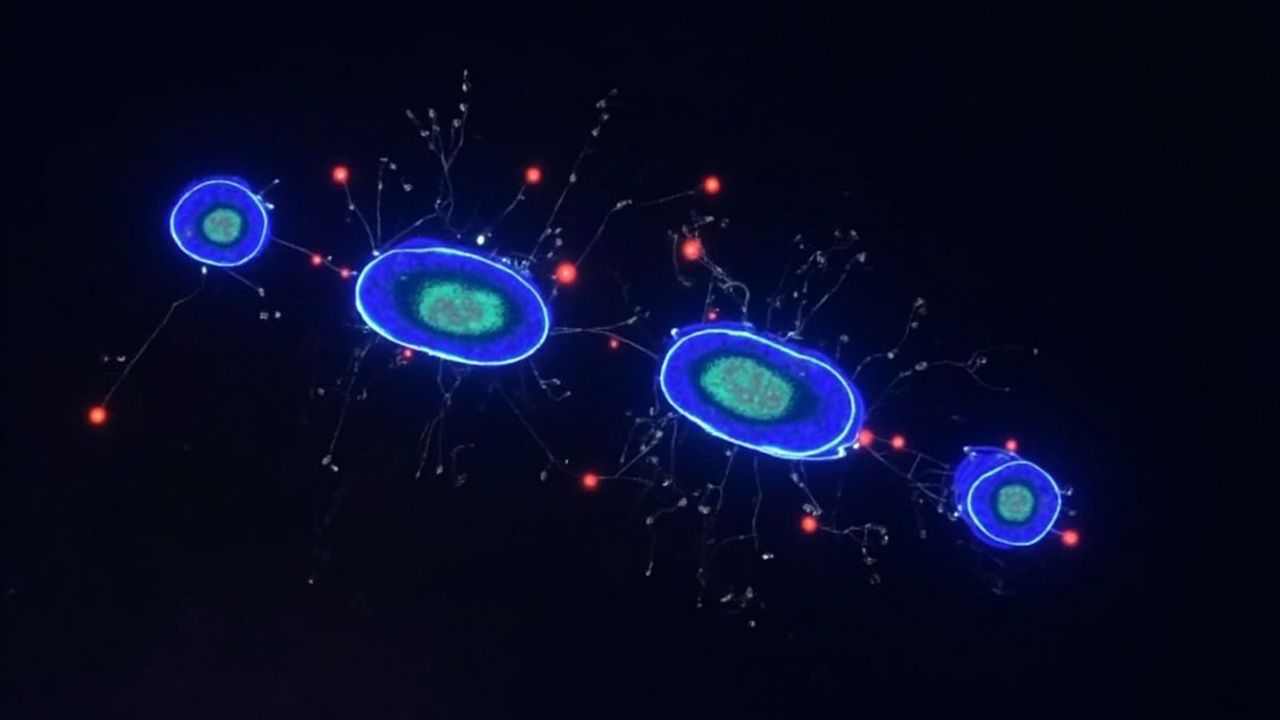
Instead of tinkering around the edges, the Swedish researchers overhauled their whole approach. They refined how they grew the cells and let them form natural 3D clusters rather than flat layers. This seemingly small change had a huge impact—suddenly the cells were way more mature and actually responded properly to glucose levels, just like real insulin cells should.
When they transplanted these cells into diabetic mice (placed them in the eye where researchers could easily observe them), something remarkable happened: the mice's blood sugar gradually stabilized. The cells didn't just survive—they kept working for months.
Why This Actually Matters
What's genuinely exciting here isn't just that it worked in mice. (We've seen that before.) It's that the researchers solved multiple longstanding problems at once:
Better cell quality - The new method actually produces the right cell types, not a messy mixture
Real maturity - These cells aren't half-baked. They respond to glucose like they're supposed to
Consistency - This works across different stem cell lines, meaning it's reliable and scalable
Patient-specific potential - You could theoretically use a patient's own cells, which means their immune system might accept them without rejection
So When Can Diabetics Get This Treatment?
Here's where I have to be honest: we're not there yet. The researchers themselves say this is a stepping stone toward clinical translation—that's science-speak for "we need to do human trials now." Other teams are already testing stem cell therapies in actual patients, but those treatments haven't been perfect. This new breakthrough addresses why those early attempts ran into trouble.
Think of it like this: we finally have the blueprint for building reliable insulin-producing cells. Now comes the harder part—making sure they work safely in humans, figuring out the right dose, preventing rejection, and jumping through all the regulatory hoops. That usually takes years.
The Big Picture
What I love about this research is that it's not just incremental. The scientists didn't just tweak someone else's method—they went back to basics and rebuilt the process from the ground up. That's how real breakthroughs happen.
For the millions of people managing type 1 diabetes every single day, this research is one more piece of hope. We're getting closer to a future where instead of injecting insulin, people might get a personalized cellular therapy that actually replaces what their immune system destroyed. That's worth getting excited about.
The study was published in Stem Cell Reports and involved researchers from Karolinska Institutet and KTH Royal Institute of Technology, with funding from organizations including the Swedish Research Council, the Novo Nordisk Foundation, and the European Research Council.